|
The nutritional supplements market in Germany is witnessing a surge in trends and opportunities, driven by changing consumer preferences and a heightened awareness of health and wellness. These trends not only reflect the current state of the market but also present promising opportunities for businesses looking to capitalize on the growing demand for nutritional supplements.
Germany nutritional supplements market was valued at US$ 4.86 Billion in 2023, and is expected to exhibit a CAGR of 3.3%, over the forecast period (2023-2030) to reach US$ 6.28 Billion by 2030. One notable trend is the rise of functional foods and beverages infused with nutritional supplements. Consumers are increasingly seeking convenient and enjoyable ways to incorporate supplements into their daily routines. This has led to the development of innovative products such as fortified snacks, protein-enriched beverages, and personalized nutrition solutions. Companies that can tap into this trend stand to gain a competitive edge in the market. Another significant opportunity lies in the intersection of technology and nutrition. The use of digital platforms and mobile apps for personalized health tracking and recommendations is gaining traction. Consumers are looking for seamless and tech-driven solutions to monitor their health and receive personalized advice on nutritional supplements. Companies that invest in digital health technologies and data-driven insights are well-positioned to capture this emerging market segment. The Germany Nutritional Supplements Market Share is also witnessing a growing emphasis on sports nutrition and wellness. With an increasing number of individuals adopting active lifestyles, there is a heightened demand for supplements that support physical performance, recovery, and overall well-being. Brands that can cater to this niche market with scientifically-backed products are likely to experience significant growth. The nutritional supplements market in Germany is evolving rapidly, presenting a spectrum of opportunities for businesses to thrive. Adapting to changing consumer preferences, embracing innovation, and leveraging emerging trends are essential strategies for companies seeking success in this dynamic and competitive market.
0 Comments
Patient derived xenograft (PDX) model enables the propagation of tumor tissues directly from patients into immunocompromised mice. These models help in recapitulating histopathological and genomic features of patient tumors. PDX models help to understand cancer biology, test therapeutic responses and help in developing personalized therapies. With increasing demand for personalized medicines and targeted therapies, PDX models are gaining traction as a reliable predictive model for cancer research.
The global Patient Derived Xenograft Model Market is estimated to be valued at US$ 127 million in 2024 and is expected to exhibit a CAGR of 4.0% over the forecast period 2024 to 2031, as highlighted in a new report published by Coherent Market Insights. Market Opportunity: The growing demand for personalized cancer therapies is estimated to drive the growth of patient derived xenograft model market over the forecast period. PDX models provide an opportunity to test various therapies directly on human cancer tissues with least immunogenic barrier. This allows researchers to understand drug sensitivity/resistance profile of cancer and helps in developing targeted and personalized therapies. PDX models have emerged as gold standard for oncology drug development and are widely used for preclinical testing of cancer therapeutics. Growing focus on precision medicine is expected to drive the demand for PDX models thereby fueling the growth of the market during the forecast period. Porter's Analysis Threat of new entrants: Low due to high capital requirement for R&D and manufacturing facility. Additionally, intellectual property protection also acts as a barrier. Bargaining power of buyers: Moderate as patient derived xenograft models have limited substitutes and customers have some influence on pricing. Bargaining power of suppliers: Low given the fragmented nature of suppliers and lack of forward integration. Suppliers have limited influence on pricing. Threat of new substitutes: Low as patient derived xenograft models have distinct advantage over other preclinical models in mimicking human tumor behavior. Competitive rivalry: High owing to increasing number of players and technological advancements. SWOT Analysis Strength: Patient derived xenograft models closely mimic human tumors aiding in more accurate preclinical testing. Increased reliability and predictive value compared to cell lines. Weakness: High costs associated with development and maintenance. Scaling can be challenging. Ethical concerns regarding use of human tissues. Opportunity: Growth in precision oncology and personalized medicines is driving demand. Increased funding for cancer research worldwide will boost the market. Threats: Strict regulatory guidelines for procurement and use of human tissues. Accuracy and predictability challenges limit universal acceptance. Key Takeaways The global Patient Derived Xenograft Model Market Growth is expected to witness high growth over the forecast period of 2024 to 2031. The global Patient Derived Xenograft Model Market is estimated to be valued at US$ 127 million in 2024 and is expected to exhibit a CAGR of 4.0% over the forecast period 2024 to 2031. North America currently dominates the market owing to presence of major players and higher adoption of advanced technologies. The Asia Pacific region is anticipated to grow at the fastest pace with increasing R&D spending and focus on precision medicine in countries like China and India. Key players operating in the patient derived xenograft model market are Crown Bioscience Inc., WuXi AppTec, Champions Oncology, The Jackson Laboratory, ONCOMEDIC, Horizon Discovery Group plc., Urolead, Xenopat, Champions Biotechnology, and Aragen Bioscience. For more insights, read- https://www.newsstatix.com/patient-derived-xenograft-model-market-trends-size-and-share-analysis/ For more details on the report, Read- https://dailynewsmotion.weebly.com/report-blog/plant-based-meat-market-is-estimated-to-witness-high-growth-owing-to-opportunity The global tissue plasminogen activator market is primarily engaged in the manufacturing and development of biotechnology drugs required for the treatment of thrombotic conditions such as myocardial infarction, deep vein thrombosis, and pulmonary embolism. Tissue plasminogen activator or tPA helps in dissolving blood clots by converting plasminogen into plasmin, a proteolytic enzyme. It has emerged as the standard of care for thrombolytic therapies post acute ischemic stroke and myocardial infarction. With high prevalence of cardiovascular diseases and rising ischemic strokes, the demand for tPA drugs has been consistently rising over the years.
The global tissue plasminogen activator Market is estimated to be valued at US$ 2.58 Bn in 2023 and is expected to exhibit a CAGR of 6.1% over the forecast period 2023 to 2030, as highlighted in a new report published by Coherent Market Insights. Market Dynamics: One of the key drivers for the growth of the tissue plasminogen activator market is the high prevalence of thrombotic conditions globally. As per estimates by WHO, cardiovascular diseases are the leading cause of death worldwide, taking an estimate of over 17.9 million lives each year. Ischemic heart disease and strokes alone accounted for 85% of all cardiovascular disease deaths. With rising risk factors such as obesity, sedentary lifestyle and expanding geriatric population, the incidences of myocardial infarctions, ischemic strokes, and deep vein thrombosis are expected to surge significantly thereby propelling demand for tPA drugs in thrombolytic therapies over the forecast period. Additionally, ongoing clinical trials exploring new therapeutic applications and delivery modes of tPA are anticipated to further augment the market growth. For instance, studies are being conducted to evaluate the efficacy and safety of tPA in extending the treatment window for ischemic stroke from 4.5 hours to 9 hours post onset of symptoms. SWOT Analysis Strength: Tissue plasminogen activator has high specificity in dissolving blood clots compared to other thrombolytic drugs. It can effectively treat pulmonary embolism and ischemic stroke when administered within a 4.5 hour window from symptom onset. Tissue plasminogen activator is administered intravenously which allows for targeted delivery and immediate impact. Weakness: Tissue plasminogen activator has a narrow therapeutic window and risks hemorrhage if given outside the approved time window. The risks associated with bleeding has led to under-utilization of the drug. Additionally, tissue plasminogen activator needs to be refrigerated during transportation and storage which adds to logistical challenges. Opportunity: Expanding approved treatment window and development of more convenient delivery formulations like inhaled versions can help address current challenges and expand the potential patient pool. Increasing public awareness about time-sensitive nature of stroke and importance of rapid treatment with tissue plasminogen activator can boost its utilization rates. Threats: Competition from newer thrombolytic drugs with wider safety margins poses pricing pressure on tissue plasminogen activator. Stringent regulatory pathways for new drugs limits frequent product upgrades for tissue plasminogen activator. Key Takeaways The global Tissue Plasminogen Activator Market Share is expected to witness high growth over the forecast period owing to increasing incidence of stroke, pulmonary embolism and growing research focus on expanding tissue plasminogen activator's therapeutic applications. The global tissue plasminogen activator Market is estimated to be valued at US$ 2.58 Bn in 2023 and is expected to exhibit a CAGR of 6.1% over the forecast period 2023 to 2030. Geographical analysis indicates that North America currently dominates the market due to supportive reimbursement policies and ability to afford premium priced drugs. However, Asia Pacific region is expected to see rapid adoption of tissue plasminogen activator driven by rising healthcare awareness, better access and growing disease prevalence due to lifestyle changes. Key players operating in the tissue plasminogen activator market are Boehringer Ingelheim, Emcure Pharmaceuticals, Genentech, and Mochida Pharmaceutical. Some of the strategies adopted by leading companies include new product approvals for alternative routes of administration and expanded treatment window approvals to address current challenges. Collaborations with research institutes are ongoing to further optimize tissue plasminogen activator formulations. For more insights, read- https://www.pressreleasebulletin.com/tissue-plasminogen-activator-market-trends-size-and-share-analysis/ Orthodontics involves the treatment of misaligned teeth and jaws, as well as dental irregularities. It involves the use of braces, retainers, and other orthodontic appliances to align and straighten teeth to achieve an ideal occlusion and aesthetics. The orthodontic devices market involves product categories such as clear aligners, fixed braces, and lab-fabricated braces. With advancements in 3D printing technology, customized clear aligners and braces can now be produced. This provides advantages such as reduced treatment time and chair time at clinics. Also, the COVID-19 pandemic has increased the adoption of telehealth services and remote orthodontic treatments through teleconsultations.
The global orthodontics market is estimated to be valued at US$ 5.06 Bn in 2023 and is expected to exhibit a CAGR of 13% over the forecast period 2023 to 2030, as highlighted in a new report published by Coherent Market Insights. Market Dynamics: Increasing Investments in Telehealth Services and In-Home Orthodontic Procedures: The key driver fueling the growth of this market is the increasing investments made by orthodontic product manufacturers and service providers towards developing innovative telehealth and remote monitoring solutions. This allows dentists and orthodontists to provide consultations and aligners fitting and monitoring services via video conferencing without requiring in-person visits. Companies are also offering mail-order impression kits and doorstep delivery of products. These trends have boosted the accessibility and affordability of orthodontic treatments. Advancements in 3D Printing: Additionally, ongoing research and development activities towards enhancing 3D printing and digital technologies for customized orthodontic treatment solutions are also propelling the industry growth. 3D printing helps in digitally designing precision-fit braces and aligners, leading to higher success and compliance for treatment plans. It reduces errors, production time, and costs of fabricating oral devices. The integration of 3D scanning, CAD/CAM software, and 3D printing technologies provide customized solutions tailored as per the patient's oral structure. SWOT Analysis Strength: Orthodontics procedures help patients improve their oral health, confidence, and quality of life through properly aligned teeth and an improved smile. These procedures are less invasive than other dental procedures and utilize removable or fixed orthodontic appliances to gradually adjust teeth positions. Many orthodontic treatment plans are designed to be completed in 1-2 years. Weakness: Orthodontic treatments can be expensive, with total costs often ranging between $5,000-$8,000 depending on treatment complexity. Some patients may not be able to afford the upfront costs or monthly payments required. Additionally, orthodontic appliances require diligent cleaning and adjustments over an extended period of time, which some patients find inconvenient. Opportunity: Rising income levels and dental insurance coverage are increasing accessibility and affordability of orthodontic care globally. Rapid adoption of new orthodontic technologies like clear aligners, lingual braces, and 3D imaging systems are fueling market growth. In addition, a growing focus on aesthetics is driving demand for orthodontic treatments among both children and adults. Threats: Economic slowdowns could negatively impact discretionary spending on orthodontic treatments. Alternative straightening methods available without orthodontist involvement also present competition. Additionally, the COVID-19 pandemic temporarily disrupted orthodontic practices and patient visits in 2020-21. Key Takeaways The global Orthodontics Market Growth is expected to witness high growth over the forecast period of 2023 to 2030 supported by rising accessibility of these procedures. The market size is projected to reach US$ 5.06 billion by 2024. Regional analysis: North America presently dominates the orthodontics market due to high patient acceptance and regional presence of leading orthodontic products and service providers. However, Asia Pacific is expected to experience the fastest growth fueled by expanding healthcare infrastructure, large patient pools and rising medical tourism in countries like India, China, and South Korea. Key players: Key players operating in the orthodontics market include J.P. Morgan Treasury Services, Bank of America Merrill Lynch, Citibank, Wells Fargo, HSBC Global Banking and Markets, BNP Paribas, Deutsche Bank, PNC Bank, Barclays, and U.S. Bank. These firms provide financing solutions for orthodontic practices and patients to facilitate treatment access Human insulin drugs such as rapid-acting insulin, long-acting insulin, and premixed insulin are used for diabetes treatment. These drugs help control blood glucose levels in the body. Rising obesity levels have increased the risk of diabetes globally. According to the International Diabetes Federation, over 537 million adults are living with diabetes worldwide in 2021, and the number is expected to rise to 643 million by 2030 and 783 million by 2045. The increasing prevalence of diabetes is anticipated to boost the demand for effective treatment options like human insulin drugs over the forecast period.
The global human insulin drug market is estimated to be valued at US$ 49411.32 Mn in 2023 and is expected to exhibit a CAGR of 6.1% over the forecast period 2023 to 2030, as highlighted in a new report published by Coherent Market Insights. Market Dynamics: One of the key drivers for the growth of the human insulin drug market is the rising prevalence of diabetes. As mentioned above, obesity levels have increased considerably in recent years. Obesity is a major risk factor for type 2 diabetes. According to the World Health Organization (WHO), in 2016, around 1.9 billion adults aged 18 years and older were overweight, and out of these over 650 million adults were obese. If the obesity trends continue, nearly half of the world's adult population is projected to be overweight or obese by 2030. The increasing obese population subsequently raises the risk of diabetes, driving the demand for diabetes management drugs like human insulin. Another driver for the market growth is continuous technological advancements in insulin delivery devices and treatments. Research is focused on developing new generations of rapid-acting and long-acting insulin analogs that can provide the smooth control of blood glucose levels similar to healthy pancreas. Companies are engaged in developing artificial pancreas devices and non-invasive insulin delivery technologies to make treatment more convenient for people with diabetes. These innovations are expected to boost the adoption of human insulin drugs over the projected period. SWOT Analysis Strength: The human insulin drug market has high unmet needs as diabetes is increasing globally. The presence of several major players in this market ensures constant innovation and development of new and advanced products. Reimbursements and awareness programs by governments further boost the adoption of human insulin drugs. Weakness: The production of human insulin requires complex extraction and purification processes from animal sources which increases costs. Biosimilar versions also pose pricing competition to innovator brands. Side effects associated with insulin therapy and needle-phobia among patients are other challenges. Opportunity: Rising geriatric population susceptible to diabetes coupled with obese population growth present new patient pools. The development of alternative delivery methods like inhaled insulin reduces pain and vulnerability of injections. Partnerships between players to reach untapped lower-income markets can spur growth. Threats: Stringent regulations for approval of new drugs and changes in reimbursement policies impacts revenues. The development of alternate treatment options threatens the demand for insulin drugs. Patent expiries of key brands enable competition from biosimilars. Key Takeaways The global human insulin drug market is expected to witness high growth. Regional Analysis: North America currently dominates due to the large diabetic patient pool and favorable insurance coverage in the region. However, Asia Pacific is poised to be the fastest-growing market owing to rising diabetes prevalence, growing healthcare sector and large untapped opportunities. Key players operating in the Human Insulin Drug Market Growth are Ecolab Inc., Rollins Inc., Rentokil Initial Plc., ServiceMaster Company, LLC, Massey Services Inc., Arrow Exterminators Inc., Sanix Incorporated, Asante Inc., Dodson Pest Control, Inc., Target Specialty Products, Pelsis Ltd., Killgerm Ltd., WinField Solutions, LLC, and Univer Inc. They are focused on strengthening their product portfolios and market presence through mergers, collaborations and new product launches. For more insights, read- https://www.pressreleasebulletin.com/human-insulin-drug-market-trends-size-and-share-analysis Injectable drugs are drugs formulated to be administered parenterally, with routes of administration including intravenous (IV), intramuscular (IM), subcutaneous (SC) and others. Injectable formulations bypass the gastrointestinal tract and liver, allowing for more rapid absorption and systemic effect. They are commonly used in hospital and clinical settings for the treatment of acute and chronic conditions that require rapid onset of therapeutic drug levels or when oral administration is not possible. Some key therapeutic classes treated via injectable routes include analgesics, anesthetics, anti-infectives, hormones, immunomodulating biologics and supportive care drugs. The global injectable drugs market has evolved tremendously in recent years, driven by rising adoption of biologics and the demand for convenient drug administration with enhanced efficacy.
The global Brazil Injectable Drugs Market for Hospitals & Ambulatory Settings is estimated to be valued at US$ 9353.45 Mn in 2023 and is expected to exhibit a CAGR of 7.8% over the forecast period 2023 to 2030, as highlighted in a new report published by Coherent Market Insights. Market Dynamics: One of the major drivers fueling growth of the Brazil Injectable Drugs Market for Hospitals & Ambulatory Settings is the growing preference for injectable formulations. Parenteral administration provides direct access to systemic circulation, allowing injections to demonstrate a rapid onset of action as compared to oral formulations. This is a major advantage for acute treatments and urgent medical conditions. Further, injections allow for direct delivery of drugs to target sites bypassing the gastrointestinal tract and first-pass hepatic metabolism, enabling higher bioavailability for certain drugs. Some new biological drugs can only be effectively delivered via injection owing to factors such as instability in the GI tract, large molecular size preventing absorption from intestine and site-specific action in body. Rising uptake of specialty biologics in therapeutic areas like oncology, rheumatology and neurology is hence expected to propel the adoption of injectable drugs in Brazil over the forecast period. SWOT Analysis Strength: The Brazil injectable drugs market for hospitals and ambulatory settings benefits from a well-established healthcare infrastructure and universal healthcare access. The rising prevalence of chronic diseases like cancer, diabetes, and cardiovascular diseases has increased the demand for injectable drugs. Reimbursements and favorable government initiatives are driving the growth of the market. Weakness: High costs associated with R&D, manufacturing, and quality control of injectable drugs put pricing pressure on players. Lack of skilled pharmacists to administer injectable medications is another challenge faced by the market. Opportunity: Growing geriatric population suffering from multiple conditions creates a vast opportunity for novel injectable drugs. Increasing adoption of self-injectable drugs offers potential for market expansion. Partnerships between players can help develop cost-effective drugs for underserved patient segments. Threats: Stringent regulations regarding drug development, manufacturing, and quality make market entry difficult. Shortage of essential injectable drugs poses a threat if not addressed properly. Patent expirations of blockbuster drugs impact market revenues of existing players. Key Takeaways The Brazil Injectable Drugs Market for Hospitals & Ambulatory Settings Growth is expected to witness high growth driven by rising disease burden, growing healthcare expenditures, and availability of reimbursements. The market size is forecast to reach US$ 9353.45 Mn by 2024, expanding at a CAGR of 7.8% during the forecast period of 2023 to 2030. Regional analysis: Brazil dominates the injectable drugs market in Latin America with more than 80% share. Rising access to universal healthcare and presence of major players have supported market growth in the country. The government is taking initiatives to boost local manufacturing and imports of essential drugs. Key players: Key players operating in the Brazil Injectable Drugs Market for Hospitals & Ambulatory Settings are Danaher Corporation, Agilent Technologies, Thermo Fisher Scientific, Merck KGaA, Stratedigm Inc., Sysmex Partec GmbH, Bio-Rad Laboratories, Miltenyi Biotec, Predictive Health Diagnostics Company, Inc., ZEISS Group International, Lambert Instruments, Nikon Instruments Inc., Leica Microsystems, and Nikon Corporation. Theyare focusing on partnerships, new product launches and expanding manufacturing facilities to strengthen market position. For more insights, read- https://www.pressreleasebulletin.com/brazil-injectable-drugs-market-for-hospitals-ambulatory-settings-trends-size-and-share-analysis The smart hospital concept makes use of advanced medical devices and equipment with integrated electronics, sensor and networking capabilities to digitize patient health records, monitor vital signs, and enable remote diagnosis and treatment. It connects all departments including emergency and outpatient care, pharmacy, laboratory, and radiology on a single digital platform for better healthcare administration and treatment. This leads improved clinical outcomes and safety of patients along with operational efficiency in dealing with the burgeoning healthcare needs worldwide. The global smart hospitals market is estimated to be valued at US$41.00 Bn in 2023 and is expected to exhibit a CAGR of 6.7% over the forecast period 2023 to 2030, as highlighted in a new report published by Coherent Market Insights.
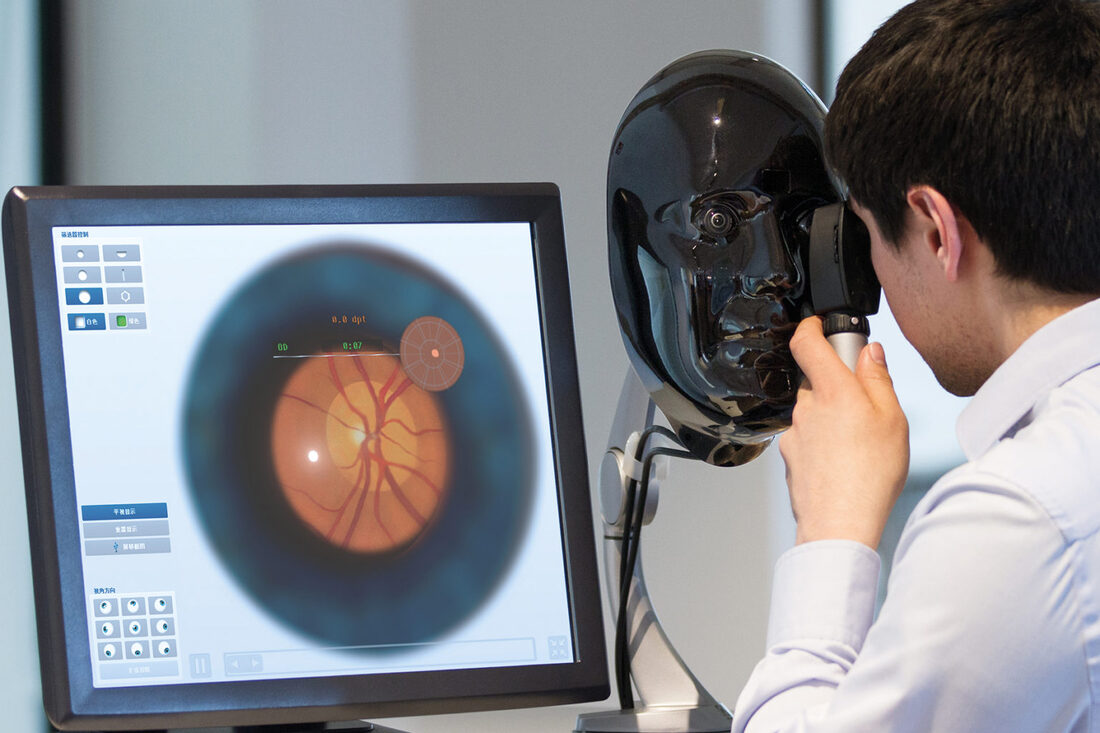
Market Dynamics: Growing digitization across the healthcare industry along with rising investments in building connected healthcare infrastructure by both private and public entities are the key drivers aiding the growth of smart hospitals market. The influx of latest digital technologies from remote patient monitoring solutions, electronic medical records, telehealth devices to blockchain and artificial intelligence are enabling hospitals to better manage large patient loads, reduce medical errors and enhance doctor-patient experience. Furthermore, integration of IoT-based devices in surgical processes, drug management and wearable health trackers in smart hospitals allows for continuous real-time monitoring of vitals and automated exchange of medical histories between stakeholders which boosts treatment quality. SWOT Analysis Strength: Smart hospitals leverage technologies like IoT, AI and cloud to improve efficiency. Digital patient records allow healthcare providers real-time access to complete medical histories. Remote patient monitoring enables early detection of health issues. Weakness: High upfront infrastructure costs associated with digital transformation strain healthcare budgets. Lack of technical skills among clinical staff impedes the use of advanced technologies. Ensuring privacy and security of sensitive patient data is challenging. Opportunity: Growing geriatric population and rising chronic diseases drive demand for telehealth and remote care services. Adoption of 5G networks facilitates development of new digital health solutions. Government initiatives promote large-scale deployment of eHealth programs. Threats: Technical glitches and system errors compromise care quality and patient safety. Hackers targeting vulnerabilities in connected medical devices pose cybersecurity risks. Physicians resistant to change may be reluctant to adopt digital practices. Key Takeaways The global Smart Hospitals Market Growth is expected to witness high growth. Regional analysis: Asia Pacific region is poised to be the fastest growing market for smart hospitals during the forecast period. Countries like China, India and Japan are increasingly investing in digital healthcare infrastructure to meet the needs of their large population bases. Key players related content comprises Key players operating in the smart hospitals market are Silvert’s Adaptive Clothing & Footwear, Izzy Camilleri, NBZ Apparel International, Able2Wear, Adaptions by Adrian, Professional Fit Clothing, Adaptive Clothing Showroom, Creation Confort, Buck & Buck, PVH Corp. and Lycra Company. These companies are focusing on developing AI-powered clinical solutions, next-gen telehealth services and cloud-based hospital information systems. Partnerships with regional healthcare providers will help them expand into new markets. For more insights, read- https://www.pressreleasebulletin.com/smart-hospitals-market-trends-size-and-share-analysis/ A laser indirect ophthalmoscope (LIO) allows physicians to perform detailed examinations of the interior structures of the eye, such as the retina, optic disc, and macula. It provides a high-quality magnified view of the fundus and employs a laser light source rather than traditional halogen bulbs. LIOs offer several benefits over conventional models, including greater illumination and better resolution. They are widely used in screening programs and for diagnosing conditions like glaucoma, macular degeneration, and diabetic retinopathy.
The global laser indirect ophthalmoscope market is estimated to be valued at US$ 116.01 Million in 2023 and is expected to exhibit a CAGR of 4.4% over the forecast period 2023 to 2030, as highlighted in a new report published by Coherent Market Insights. Market Dynamics: The rising prevalence of eye diseases remains the primary driver for the laser indirect ophthalmoscope market. According to estimates by the World Health Organization (WHO), at least 2.2 billion people have a vision impairment or blindness, with eye conditions like cataracts, glaucoma, and diabetic retinopathy being among the leading causes worldwide. Laser indirect ophthalmoscopes offer distinct advantages in screening for and managing these chronic illnesses compared to conventional models. Furthermore, ongoing technological innovations focused on improving image quality, reliability, and ease-of-use have increased the adoption of LIOs among eye care professionals. SWOT Analysis Strength: The laser indirect ophthalmoscope market offers non-invasive procedures that allow examination of the retina without requiring pupil dilation. This enables doctors to efficiently diagnose diseases. The laser systems offer high-quality images and versatility for addressing different eye conditions. They provide digital documentation and imaging capabilities for record-keeping. Weakness: The laser indirect ophthalmoscope equipment is expensive to purchase and maintain over the long run. This poses a barrier for smaller clinics and practices. Some older patients may find it difficult to remain still during examination using these systems. Opportunity: Rising incidence of eye diseases worldwide due to increasing life expectancy and growing diabetic population present significant opportunities. Development of integrated systems combining indirect ophthalmoscopy with other modalities can widen the scope of applications. Incorporating advanced features such as OCT and fundus autofluorescence expands clinical utility. Threats: Reimbursement challenges and lack of insurance coverage for digital equipment in some regions hamper adoption rates. Availability of cheaper substitute examination methods act as a hindrance. Economic slowdowns can negatively impact the demand for such capital equipment. Key Takeaways The global Laser Indirect Ophthalmoscope Market Growth is expected to witness high growth. North America dominates currently due to supportive reimbursement scenario and availability of advanced healthcare infrastructure. Growing population dealing with chronic diseases drive the demand for ophthalmic diagnostic equipment in the region. Asia Pacific is poised to emerge as the fastest growing market owing to rising healthcare spending, increasing awareness about eye care, and rapid expansion of optical retail chains. Key players operating in the laser indirect ophthalmoscope market are Ellex Medical Lasers Ltd, Iridex Corporation, Alcon, Carl Zeiss Meditec AG, Lumenis Ltd., Topcon Medical Laser System, Nidek Co. Inc., Ziemer Ophthalmic Systems AG, Heine Optotechnik GmbH, and Keeler Ltd. The major companies focus on new product introductions and expanding presence across global markets. Partnerships with eye hospitals and clinics help raise brand visibility and capture untapped opportunities. Aggressive marketing campaigns spread awareness regarding benefits of digital equipment. The global laser indirect ophthalmoscope market is fragmented with the presence of both international and regional vendors. Differentiation is achieved through technologically advanced products, competitive pricing, and robust after-sales support. Manufacturers face the dual challenges of meeting stringent regulatory norms as well as catering to shifting demand towards multifunctional devices. Those able to align their strategies according to evolving industry trends are expected to gain competitive advantage. For more insights, read- https://www.pressreleasebulletin.com/laser-indirect-ophthalmoscope-market-trends-size-and-share-analysis/ Laboratory proficiency testing involves evaluating a laboratory's performance using inter-laboratory comparisons. Laboratories performing clinical or environmental testing enroll in proficiency testing programs provided by third-party proficiency testing providers to test the accuracy of their testing methods and identify potential problems. The testing programs provide samples to laboratories for analysis and evaluation. It ensures laboratories are actively monitoring performance and identifies needs for quality improvement. With growing regulatory focus on quality control and assurance of laboratory test results, demand for proficiency testing services is on the rise.
The global laboratory proficiency testing market is estimated to be valued at US$ 1.29 Bn or Mn in 2023 and is expected to exhibit a CAGR of 2.6% over the forecast period 2023 to 2030, as highlighted in a new report published by Coherent Market Insights. Market Dynamics: The increasing focus on quality control and assurance of laboratory test results is a key driver boosting growth of the global laboratory proficiency testing market over the forecast period. Regulatory bodies across countries have mandated regulations for laboratories to undergo regular proficiency testing in order to obtain accreditation and certifications required to perform clinical or environmental tests. This ensures reliability of test results and accuracy of testing methods adopted by laboratories. Additionally, the integration of artificial intelligence technologies by proficiency testing providers is also expected to offer new growth opportunities over the coming years. AI allows for faster, more efficient and cost-effective proficiency testing services through automation of processes and online proficiency testing programs. However, challenges associated with maintaining supply chain operations during the ongoing COVID-19 pandemic may hinder market growth to some extent over the short term. SWOT Analysis Strength: The Laboratory Proficiency Testing Market has strong presence of key players such as Mason Corporation and Ace Chemical Company. These companies have extensive experience providing proficiency testing services to laboratories globally. Furthermore, providing accurate testing results is crucial for healthcare services so demand is consistent. Weakness: Small testing laboratories may find proficiency testing programs too expensive. Complying with different regulatory standards also increases costs for market players. Scheduling proficiency testing can also disrupt the regular operations of some smaller clinical labs. Opportunity: The market is expected to grow in developing regions like Asia Pacific and Latin America owing to increasing investments in healthcare infrastructure. Growing awareness among laboratories about the benefits of proficiency testing also presents an opportunity. Market players can further expand service offerings such as online reporting of results. Threats: Presence of local testing service providers poses threat in price competition. Strict regulatory norms in healthcare may increase compliance costs for companies. Economic slowdowns can negatively impact investments in proficiency testing programs by various end users. Key Takeaways The global Laboratory Proficiency Testing Market Growth is expected to witness high growth. Regional analysis: Asia Pacific witnessing the fastest growth in the market. With rising healthcare infrastructure and growing patient pool, countries like China, India are expected to offer lucrative opportunities. Key players operating in the Laboratory Proficiency Testing Market are Mason Corporation, Ace Chemical Company, City Chemicals LLC, TIB Chemicals AG, Pfaltz & Bauer Inc., GFS Chemicals, Showa America, S V Plastochem Private Limited, Westman Chemicals Pvt, William Blythe, Haihang Industry Co.,Ltd, and Strem Chemicals. These companies are focusing on strategic partnerships and collaborations to strengthen their presence across different regions. For more insights, read- https://www.pressreleasebulletin.com/laboratory-proficiency-testing-market-trends-size-and-share-analysis/ Scopolamine is an alkaloid drug derived from plants belonging to the family Solanaceae, which provides relief from motion sickness. It works by blocking signals in the brain that cause nausea and vomiting. Scopolamine is used to treat and prevent motion sickness, postoperative nausea and vomiting, nausea associated with cancer chemotherapy and gastroenteritis.
The global scopolamine API market is estimated to be valued at US$ 308.49 Mn in 2023 and is expected to exhibit a CAGR of 10% over the forecast period 2023 to 2030, as highlighted in a new report published by Coherent Market Insights. Market Dynamics: Growth in the global scopolamine API market is majorly driven by increasing applications of scopolamine API in treatment of motion sickness as well as nausea and vomiting associated with cancer chemotherapy and postoperative nausea and vomiting. Scopolamine is considered as the gold standard drug for prevention of motion sickness owing to its strong antiemetic effects and longer duration of action. According to various published studies, scopolamine patches are more than 80% effective in preventing motion sickness. Scopolamine patches and gums have become popular OTC medication for motion sickness. Thus, increasing incidence of motion sickness during travel via road, rail or sea is expected to drive the demand for scopolamine API over the forecast period. SWOT Analysis Strength: The Scopolamine API market is expected to grow at a CAGR of 10% during the forecast period. Scopolamine is used to treat motion sickness and post-operative nausea. The increasing demand for pharmaceutical products is driving market growth. The rising prevalence of cancer and neurological disorders is fuelling demand for scopolamine-based drugs. Weakness: Stringent regulations associated with API manufacturing may hamper market growth. High costs associated with R&D activities can negatively impact small players. Opportunity: Growing geriatric population suffering from neurological disorders creates lucrative opportunities. Increasing healthcare expenditure in emerging nations provides potential opportunities. Threats: Availability of alternative therapies and substitutes poses a threat. Rising environmental concerns associated with API production threats market growth. Key Takeaways The global Scopolamine API Market Growth is expected to witness high growth. The global scopolamine API Market is estimated to be valued at US$ 308.49 Mn in 2023 and is expected to exhibit a CAGR of 10 % over the forecast period 2023 to 2030. North America is currently the largest market for scopolamine API due to rising prevalence of motion sickness and cancer. The dominant market share can be attributed to growing healthcare expenditure, availability of advanced healthcare facilities, and presence of prominent market players in the region. Asia Pacific is expected to be the fastest-growing regional market owing to rising geriatric population, growing healthcare spending, and increasing prevalence of cancer and neurological disorders. Moreover, increasing investments by global market players are driving market growth in Asia Pacific region. Key players operating in the scopolamine API market include Daikin Industries Limited, Viessmann Group, NIBE Group, Danfoss Group Global, Stiebel Eltron Group, Mitsubishi Electric Corporation, United Technologies Corporation, Panasonic Corporation, Glen Dimplex Group, The Bosch Group. The market is dominated by global players accounting for more than 50% of the total market share. Strategic collaborations, mergers, acquisitions, new product launches, and regional expansion are common strategies adopted by these companies to gain higher market shares and strengthen their product portfolio. For more insights, read- https://www.pressreleasebulletin.com/scopolamine-api-market-trends-size-and-share-analysis/ |
AuthorWrite something about yourself. No need to be fancy, just an overview. Archives
February 2024
Categories
All
|



 RSS Feed
RSS Feed
